Science Links:
| sample-muscles-worksheets.pdf | |
| File Size: | 1832 kb |
| File Type: | |
https://www.ducksters.com/science/simple_machines.php
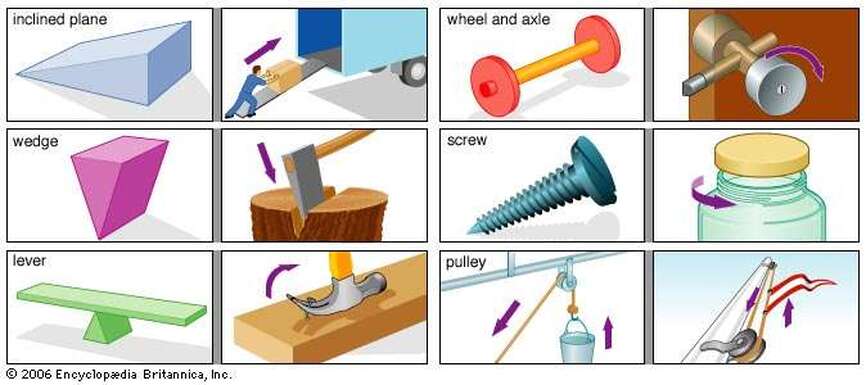
Criteria for Simple Machines Comic:
- Good effort shown
- Outlined in Sharpie
- Colourful
- Includes a bold title
- Elements of a comic are included (cartoon characters, speech and think bubbles, captions, sound effect)
- Demonstrates an understanding of simple machines
BORAX CRYSTALS - HOW DOES IT WORK?
When you mixed the borax and water, you created a suspension of borax. A suspension is a mixture that contains solid particles large enough to make the liquid appear cloudy or murky. By mixing the borax into hot water, instead of room temperature or cold water, the borax can stay suspended much longer. Very hot water can hold much more dissolved borax than cold water. Hot water molecules are moving very fast and are spread way out which makes space available for more borax to dissolve into it. As the mixture cools, the water molecules slow down and move closer together. That means there’s less room for the dissolved borax and it begins to fall out of the water.
As the borax settles out out of the cooling suspension due to gravity, it bonds with other borax on nucleation sites (bumps, tiny cracks, impurities, etc. in the container) and begins to form seeds for further crystallization. Crystals come together in specific, repeated patterns due to the shape of the molecules forming them. You’ll see this crystallization on the bottom and sides of the container, on the string hanging from the pencil, and on the pipe cleaner arms of the snowflake. The borax continues to fall and crystalize on top of the snowflake and on top of other borax crystals until you pull it out of the water the next morning.
When you mixed the borax and water, you created a suspension of borax. A suspension is a mixture that contains solid particles large enough to make the liquid appear cloudy or murky. By mixing the borax into hot water, instead of room temperature or cold water, the borax can stay suspended much longer. Very hot water can hold much more dissolved borax than cold water. Hot water molecules are moving very fast and are spread way out which makes space available for more borax to dissolve into it. As the mixture cools, the water molecules slow down and move closer together. That means there’s less room for the dissolved borax and it begins to fall out of the water.
As the borax settles out out of the cooling suspension due to gravity, it bonds with other borax on nucleation sites (bumps, tiny cracks, impurities, etc. in the container) and begins to form seeds for further crystallization. Crystals come together in specific, repeated patterns due to the shape of the molecules forming them. You’ll see this crystallization on the bottom and sides of the container, on the string hanging from the pencil, and on the pipe cleaner arms of the snowflake. The borax continues to fall and crystalize on top of the snowflake and on top of other borax crystals until you pull it out of the water the next morning.
|
Before
The Borax has been dissolved in the jar to make a suspension. When the Borax was poured into the jar it made the water murky and settled at the bottom. How did hot water and stirring make the Borax dissolve faster? |
After
You can see crystals forming on the snowflake, string and bottom of the jar. Why is it important that the snowflakes not touch the other snowflakes or the jar? |

Water molecules pull the sodium and chloride ions apart, breaking the ionic bond that held them together. After the salt compounds are pulled apart, the sodium and chloride atoms are surrounded by water molecules, as this diagram shows. Once this happens, the salt is dissolved, resulting in a homogeneous solution.